2529. Cordycepin
Nomenclature
Description and references
First reported nucleoside antibiotic. Isoln from culture fluids of Cordyceps militaris (Linn.) Link: K. G. Cunningham et al., J. Chem. Soc. 1951, 2299; N. M. Kredich, A. J. Guarino, Biochim. Biophys. Acta 41, 363 (1960). Proposed structure: H. R. Bentley et al., J. Chem. Soc. 1951, 2301. Identity with 3′-deoxyadenosine and revised structure: E. A. Kaczka et al., Biochem. Biophys. Res. Commun. 14, 456 (1964). Biosynthesis: R. Suhadolnik et al., J. Am. Chem. Soc. 86, 948 (1964). Synthesis: A. R. Todd, T. L. Ulbricht, J. Chem. Soc. 1960, 3275; W. W. Lee et al., J. Am. Chem. Soc. 83, 1906 (1961); E. Walton et al., ibid. 86, 2952 (1964); Y. Ito et al., ibid. 103, 6739 (1981). Cordycepin and cordycepin triphosphate have been used extensively in the study of messenger RNA transcription, see H. T. Shigeura, G. E. Boxer, Biochem. Biophys. Res. Commun. 17, 758 (1964); S. Penman et al., Proc. Natl. Acad. Sci. USA 67, 1878 (1970). Reviews: J. J. Fox. et al., “Nucleoside Antibiotics” in Prog. Nucleic Acid Res. Mol. Biol. 5, 258-262 (1966); A. J. Guarino, “Cordycepin” in Antibiotics I, D. Gottlieb, P. Shaw, Eds. (Springer-Verlag, New York, 1967) pp 468-480.
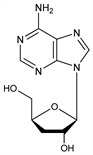
Properties
Needles from ethanol, n-butanol, n-propanol or water. mp 225-226°. [α]D20 -47°. [α]D27 -42°. uv max (ethanol): 260 nm (ε 14600). pH aq soln: 7.1.Derivative
Triphosphate.Nomenclature
Cordycepin-5′-triphosphate; 3′-deoxy ATP; 3′-deoxyadenosine-5′-(tetrahydrogen triphosphate).Description and references
Formation by conversion of 3′-deoxyadenosine in Ehrlich ascites tumor: H. Klenow, Biochim. Biophys. Acta 76, 347 (1963). Metabolism in KB cells: H. Shigeura, S. Sampson, ibid. 138, 26 (1967). Synthesis: J. J. Novak, F. Sorm, Collect. Czech. Chem. Commun. 38, 113 (1973); M. Blandin, J. Carbohydr. Nucleosides Nucleotides 3(5/6), 341 (1976).