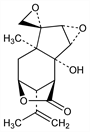
2530. Coriamyrtin
Nomenclature
Description and references
Sesquiterpene of the picrotoxane group. Main toxic principle from leaves and fruit of Coraria myrtifolia L., Coriariaceae: M. J. Riban, C. R. Hebd. Seances Acad. Sci. 57, 798 (1863) and C. japonica A. Gray, Coriariaceae: Kariyone, Sato, J. Pharm. Soc. Jpn. 50, 106 (1930); Okuda, Pharm. Bull. 2, 185 (1954). Series of articles on structure, absolute configuration, stereochemistry and derivatives: T. Okuda, T. Yoshida, Chem. Pharm. Bull. 9, 379, 404 (1961); 15, 1687, 1697 (1967). Structural relationship to tutin, q.v.: eidem, Tetrahedron Lett. 1965, 439. Biosynthetic studies: M. Biollaz, D. Arigoni, Chem. Commun. 1969, 633. Synthetic studies: K. Tanaka et al., Chem. Pharm. Bull. 31, 1943, 1958, 1972 (1983). Pharmacology: E. E. Swanson, K. K. Chen, J. Pharmacol. Exp. Ther. 57, 410 (1936). Structure-activity relationship: C. H. Jarboe et al., J. Med. Chem. 11, 729 (1968).