2552. Cotarnine
Nomenclature
CAS number: 82-54-2
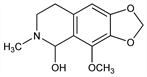
5,6,7,8-Tetrahydro-4-methoxy-6-methyl-1,3-dioxolo[4,5-g]isoquinolin-5-ol. C12H15NO4; mol wt 237.25.
C 60.75%, H 6.37%, N 5.90%, O 26.97%.
Description and references
Prepd by the oxidation of narcotine with dil nitric acid. Synthesis: Salway, J. Chem. Soc. 97, 1208 (1910). Is tautomeric. Structure studies: Small, Lutz, “Chemistry of the Opium Alkaloids,” Supplement No. 103, U.S. Public Health Reports (Washington, 1932) p 49; Schneider, Müller, Ann. 615, 34 (1958). Absorption spectrum: Csokán, Z. Anal. Chem. 124, 344 (1942).
Properties
Small needles from benzene, dec 132-133°. Sol in alcohol, chloroform, ether, benzene; slightly sol in water; sol in dil acids, in ammonia or sodium carbonate soln, but only slightly in potassium hydroxide soln. Aq or alcoholic solns are yellow.Derivative
Hydrochloride.Nomenclature
CAS number: 36647-02-6
Secalysat (Ysatfabrik). C12H15NO4.HCl; mol wt 273.71.
C 52.66%, H 5.89%, N 5.12%, O 23.38%, Cl 12.95%.
Derivative
Chloride.Nomenclature
CAS number: 10018-19-6
7,8-Dihydro-4-methoxy-6-methyl-1,3-dioxolo[4,5-g]isoquinolinium chloride; cotarnine chloride; cotarninium chloride; Stypticin (Merck & Co.). C12H14ClNO3; mol wt 255.70.
C 56.37%, H 5.52%, Cl 13.87%, N 5.48%, O 18.77%.
Properties
Dihydrate, light-yellow powder; deliquesc in moist air. Sol in about 1 part water, 4 parts alc. Keep well closed.Derivative
Phthalate.Nomenclature
CAS number: 6190-36-9
Styptol. C32H32N2O10; mol wt 604.60.
C 63.57%, H 5.33%, N 4.63%, O 26.46%.
Therapeutic Category
Hemostatic.
Keywords
Hemostatic