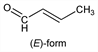Nomenclature
CAS number: 4170-30-3;
123-73-9
((E)-form)
;
15798-64-8
((Z)-form)
2-Butenal; crotonic aldehyde; β-methylacrolein.
C
4H
6O; mol wt 70.09.
C 68.54%, H 8.63%, O 22.83%.
Description and references
Commercial product is a 95:5 mixture of trans:cis isomers. Prepn: v. Auwers, Eisenlohr, J. Prakt. Chem. 82, 115 (1910);
Hibbert, J. Am. Chem. Soc. 37, 1759 (1915). Alternate synthesis: J. Smidt et al., Angew. Chem. 71,
176 (1959). Toxicity studies: H. F. Smyth, C. P. Carpenter, J. Ind. Hyg. Toxicol. 26, 269
(1944). Review of use in commercial processes: O. Horn, Ind. Eng. Chem. 51, 655-658 (1959).
Reviews: J. E. Fernandez, T. W. G. Solomons, Chem. Rev. 62, 485-502 (1962);
W. Blau et al., in Ullmann's Encyclopedia
of Industrial Chemistry vol. A8 (VCH, Weinheim,
5th ed., 1987) pp 83-89. Review of carcinogenic potential: V. J.
Feron et al., Mutat. Res. 259, 363-385 (1991); of role in DNA adduct formation:
S. S. Hecht et al., Toxicology 166, 31-36 (2001).
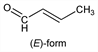
Properties
Flammable liq. Lacrimator. mp -69°; bp 102.2°; d2020 0.853; nD17.3 1.4384. Flash pt when anhydr:
13°C (55°F) (open cup). Explosive limits in air 2.95-15.5% v/v. Vapor density 2.41 (air
= 1). Heat capacity: 0.7 cal/g/°C; heat of vaporization: 123 cal/g.
Dimerizes under strong acid conditions, slowly oxidizes to crotonic
acid. Soly in water (g/100 g) at 20°: 18.1; at 5°: 19.2. Soly
of water in crotonaldehyde (g/100 g) at 20°: 9.5; at 5°: 8.0. LD50 orally in rats:
0.3 g/kg (Smyth,
Carpenter).Caution
Potential symptoms of overexposure
are irritation of eyes and respiratory system. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH
97-140, 1997) p 80.Use
Manuf of butyl alcohol, butyraldehyde, methoxybutyraldehyde,
sorbic acid, maleic acid, crotonic acid, crotyl alcohol. In polymer
chemistry: manuf of resins and polyvinyl acetals, solvent for polyvinyl
chloride, rubber antioxidant, increases rubber strength with ketones.
In prepn of insecticides and fertilizers. In production of flavors.