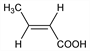Nomenclature
CAS number: 107-93-7
(2
E)-2-Butenoic acid; β-methylacrylic acid; α-crotonic acid; solid crotonic acid;
trans-crotonic acid.
C
4H
6O
2; mol wt 86.09.
C 55.81%, H 7.02%, O 37.17%.
Description and references
Has been found in clay soil in Texas; formed
during the dry distillation of wood. Obtained on a commercial scale
exclusively by oxygen- or air-oxidation of crotonaldehyde: Kennedy, US 2413235 (1946 to Shawinigan
Chem.); Leupold, PB report 70249 (1942); Matthews,
B.I.O.S. report 758 (1946). Laboratory procedure using alkaline silver
oxide: Young, J. Am. Chem. Soc. 54, 2498 (1932); from acetaldehyde and malonic acid in
pyridine: v. Auwers, Ann. 432, 46 (1923); Backer, Bloemen, Rec.
Trav. Chim. 45, 102 (1926); Florence, Bull. Soc. Chim. Fr. [4] 41, 440 (1927); Letch, Linstead, J.
Chem. Soc. 1932, 454. Toxicity study: Smyth,
Carpenter, J. Ind. Hyg. Toxicol. 26, 269 (1944). Review: W. Blau et
al., in Ullmann's Encyclopedia of Industrial
Chemistry vol. A8 (VCH, Weinheim, 5th ed.,
1987) pp 83-89.
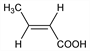
Properties
Monoclinic needles, prisms from water or ligroin.
Corrosive and combustible solid. d415 1.018; d480 0.964. mp 71.6°. bp10 80.0°; bp20 93.0°; bp40 107.8°; bp60 116.7°; bp100 128.0°; bp200 146.0°; bp400 165.5°; bp760 185.0°. nD80 1.4228. pKa (25°) 4.817. Heat of combustion: 2.00 MJ/mol.
Heat of fusion: 150.9 J/g. Soly in water (g/l) at 0°: 41.5; at
10°: 54.6; at 20°: 76.1; at 25°: 94; at 30°: 122; at 40°: 656.
Soly in ethanol at 25°: 52.5% w/w; acetone: 53.0% w/w; toluene:
37.5% w/w. LD50 orally in rats: 1.0 g/kg (Smyth, Carpenter).Derivative
Methyl ester.
C
5H
8O
2; mol wt 100.12.
C 59.98%, H 8.05%, O 31.96%.
Properties
Liq, bp 121°; d420 0.9444; nD20 1.4242.Derivative
Ethyl ester.
C
6H
10O
2; mol wt 114.14.
C 63.14%, H 8.83%, O 28.03%.
Properties
Liq, bp 138°; d420 0.9175; nD20 1.4245.Derivative
Vinyl ester.
C
6H
8O
2; mol wt 112.13.
C 64.27%, H 7.19%, O 28.54%.
Properties
Liq, bp 133°; d420 0.9410; nD20 1.450.Use
In the manuf of copolymers with vinyl acetate used
in lacquers and paper sizing; in the manuf of softening agents for
synthetic rubber. In medicinal chemistry, e.g., in the manuf of
dl-threonine, vitamin A.