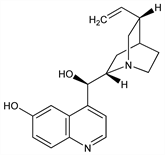
2623. Cupreine
Nomenclature
CAS number: 524-63-0
(8α,9R)-Cinchonan-6′,9-diol; hydroxycinchonidine; ultraquinine. C19H22N2O2; mol wt 310.39.
C 73.52%, H 7.14%, N 9.03%, O 10.31%.
Description and references
In bark and seeds of Remijia pedunculata Flueck., Rubiaceae. Isoln: Howard, Hodgkin, J. Chem. Soc. 41, 16 (1882). Separated from homoquinine by treatment with NaOH: Hesse, Ann. 230, 55 (1885). Reduction to hydrocupreine: Giemsa, Halberkann, Ber. 51, 1325 (1918). Conversion into cinchonidine: King, J. Chem. Soc. 1946, 523.