2721. Cycloguanil
Nomenclature
CAS number: 516-21-2
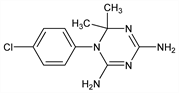
1-(4-Chlorophenyl)-1,6-dihydro-6,6-dimethyl-1,3,5-triazine-2,4-diamine; 4,6-diamino-1-(p-chlorophenyl)-1,2-dihydro-2,2-dimethyl-s-triazine; 2,4-diamino-1-p-chlorophenyl-1,6-dihydro-6,6-dimethyl-1,3,5-triazine; 1-p-chlorophenyl-2,4-diamino-6,6-dimethyl-1,6-dihydro-1,3,5-triazine; chlorazine (Russian); chlorguanide triazine; TCl; M-10580; D-20. C11H14ClN5; mol wt 251.72.
C 52.49%, H 5.61%, Cl 14.08%, N 27.82%.
Description and references
Metabolic product formed from the antimalarial drug chlorguanide: Carrington et al., Nature 168, 1080 (1951). Prepn: Modest et al., J. Am. Chem. Soc. 74, 855 (1952); Loo, ibid. 76, 5096 (1954); Carrington et al., J. Chem. Soc. 1954, 1017; Bami, J. Sci. Ind. Res. 14C, 231 (1955); Modest, J. Org. Chem. 21, 1 (1956); US 2900385 (1959 to Children's Cancer Res. Found.); Elslager, Worth, US 3074947 (1963 to Parke, Davis).
Properties
Prisms from chloroform + ether, mp 146°. uv max (water): 241 nm (log ε 4.11).Derivative
Hydrochloride.C11H14ClN5.HCl; mol wt 288.18.
C 45.85%, H 5.25%, Cl 24.60%, N 24.30%.
Properties
Prisms from water, mp 210-215°. uv max (water): 241 nm (log ε 4.12).Derivative
Dihydrochloride.C11H14ClN5.2HCl; mol wt 324.64.
C 40.70%, H 4.97%, Cl 32.76%, N 21.57%.
Properties
Crystals from acetone, mp 190-196°.Derivative
Pamoate.Nomenclature
CAS number: 609-78-9
Cycloguanil embonate; CI-501; Camolar (Parke-Davis). (C11H14ClN5)2.C23H16O6; mol wt 891.80.
C 60.61%, H 4.97%, Cl 7.95%, N 15.71%, O 10.76%.
Properties
Yellow crystals, mp 231-234°. Soly in water: 0.003%.Therapeutic Category
Antimalarial.
Keywords
Antimalarial