2853. Decitabine
Nomenclature
Description and references
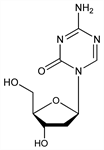
Pyrimidine analog that inhibits DNA methylation and induces differentiation of leukemic cells. Prepn: J. Pliml, F. Sorm, Collect. Czech. Chem. Commun. 29, 2576 (1964); A. Piskala, F. Sorm, in Nucleic Acid Chemistry Part 1 (Wiley, New York, 1978) pp 443-449. Improved synthesis: J. Ben-Hattar, J. Jiricny, Nucleosides Nucleotides 6, 393 (1987). Chemical stability in soln: K.-T. Lin et al., J. Pharm. Sci. 70, 1228 (1981). HPLC determn in plasma: eidem, J. Chromatogr. 345, 162 (1985). Review of pharmacology and mechanism of action: R. L. Momparler, Pharmacol. Ther. 30, 287-299 (1985); idem et al., Leukemia 11, Suppl. 1, 1-6 (1997); of clinical experience in myelodysplastic syndromes: E. Atallah et al., Expert Opin. Pharmacother. 8, 65-73 (2007).
Properties
Crystals from methanol, mp 201-202° (dec). [α]D22 +68.5° (30 min) → +57.8° (6 hr) (c = 0.5 in water). uv max (pH 7): 244 nm (log ε 3.86). Sol in DMSO; sparingly sol in water; slightly sol in methanol, methanol/water (50/50), ethanol/water (50/50). LD50 in mice (mg/kg): 190 i.p. (Momparler, 1985).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antimetabolites; Pyrimidine Analogs