2862. Deflazacort
Nomenclature
Description and references
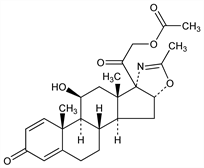
Systemic corticosteroid; oxazoline derivative of prednisolone, q.v. Prepn: G. Nathansohn, G. Winters, BE 679820; eidem, GB 1077393; eidem, US 3436389 (1966, 1967, 1969 all to Lepetit); G. Nathansohn et al., J. Med. Chem. 10, 799 (1967). Pharmacology: P. Schiatti et al., Arzneim.-Forsch. 30, 1543 (1980). Pharmacokinetics: A. Assandri et al., Eur. J. Drug Metab. Pharmacokinet. 5, 207 (1980); eidem, Adv. Exp. Med. Biol. 171, 9 (1984). Immunosuppressive activity in humans: B. H. Hahn et al., J. Rheumatol. 8, 783 (1981). Effect on human mineral metabolism: T. J. Hahn et al., Calcif. Tissue Int. 31, 109 (1980); on human glucose metabolism: P. Cavallo-Perin et al., Eur. J. Clin. Pharmacol. 26, 357 (1984). Clinical comparison with prednisone, q.v., in rheumatoid arthritis and lupus: B. Imbimbo et al., Adv. Exp. Med. Biol. 171, 241 (1984).
Properties
Crystals from acetone-hexane, mp 255-256.5°. [α]D +62.3° (c = 0.5 in chloroform). uv max (methanol): 241-242 nm (E1%1cm 352.5). LD50 orally in mice: 5200 mg/kg (Schiatti).Therapeutic Category
Anti-inflammatory; glucocorticoid.
Keywords
Glucocorticoid