2880. Delphinidin
Nomenclature
Description and references
The aglucone of delphinin: Willst"atter, Mieg, Ann. 408, 61 (1915); the aglucone of violanin: Willst"atter, Weil, ibid. 412, 178 (1917). Occurrence in plants: Acheson, Can. J. Biochem. Physiol. 37, 1 (1959). Synthesis: Pratt, Robinson, J. Chem. Soc. 127, 166 (1925); Bradley et al., ibid. 1930, 793.
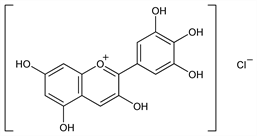
Properties
Chocolate-brown prisms or needles with metallic luster from 5% HCl. Crystallizes from water with 1, 2 or 4 mols H2O. When anhydr does not melt below 350°. Absorption max (methanolic HCl): 544 nm. Sol in methanol, ethanol, ethyl acetate.Derivative
3-Glucoside.Nomenclature
Myrtillin-a.Description and references
From the whortleberry (Vaccinium myrtillus L., Ericaceae): Willst"atter, Zollinger, Ann. 408, 83 (1915); 412, 195 (1916); from the pansy (Viola tricolor L., Violaceae): Karrer, de Meuron, Helv. Chim. Acta 16, 292 (1933). Structure and synthesis: Reynolds, Robinson, J. Chem. Soc. 1934, 1039.
Properties
Deep purple crystals from dil HCl. Absorption max (methanolic HCl): 535 nm.Derivative
3,5-Diglucoside.Nomenclature
3,5-Bis(glucosyloxy)-3′,4′,5′,7-tetrahydroxyflavylium chloride; delphin; delphoside; hyacin.Description and references
From flowers of Salvia patens Cav., Labiatae, structure and synthesis: Reynolds et al., J. Chem. Soc. 1934, 1235; from flowers of Verbena hybrida Hart., Verbenaceae: Scott-Moncrieff, Sturgess, Biochem. J. 34, 268 (1940); from grape skins: Bockian et al., J. Agric. Food Chem. 3, 695 (1955). Identity with hyacin: Saito et al., Proc. Jpn. Acad. 36, 340 (1960).
Properties
Leaflets with bronze luster from dil HCl. Absorption max (methanolic HCl): 534 nm. Practically insol in cold water, alcohol, dil acids; sol in hot dil HCl.Derivative
Compound with glucose + hydroxybenzoic acid.Nomenclature
Delphinin.Description and references
From Delphinium consolida L., Ranunculaceae: Willst"atter, Mieg, loc. cit.
Properties
Dark red-brown plates or prisms from 3% HCl. dec 200-203°. Sol in water with decompn, dil alcohol, acetone, boiling alcohol.Derivative
Compound with glucose + rhamnose + p-hydroxycinnamic acid.Nomenclature
Violanin.Description and references
From flowers of Viola tricolor L., Violaceae: Willst"atter, Weil, loc. cit. Structure: Karrer, de Meuron, loc. cit.