2899. Deoxycholic Acid
Nomenclature
Description and references
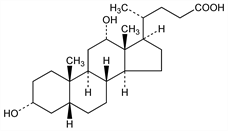
Lacks the C-7 hydroxy group of cholic acid. Occurs in bile of man, ox, sheep, dog, goat, and rabbit. Isoln: Wieland, Siebert, Z. Physiol. Chem. 262, 1 (1939). From cholic acid: Sifferd, US 2765316 (1956 to Armour). Structure and synthesis: L. F. Fieser, M. Fieser, Steroids (Reinhold, New York, 1959). Laboratory prepn from ox bile: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 361. Forms molecular coordination compds (so-called choleic acids) with many substances. Complexes wth fatty acids have been studies extensively: Sobotka, Goldberg, Biochem. J. 26, 555 (1932); Sobotka, Chem. Rev. 15, 362 (1934). Surface activity: idem, Chemistry of the Steroids, New York (1938).
Properties
Crystals from alc, mp 176-178°. Not precipitated by digitonin. [α]D20 +55° (alc). pK 6.58. Soly at 15° in water 0.24 g/l; in alcohol 220.7 g/l; in ether 1.16 g/l; in chloroform 2.94 g/l; in benzene 0.12 g/l; in acetone 10.46 g/l; in glacial acetic acid 9.06 g/l. Sol in solns of alkali hydroxides or carbonates.Derivative
Sodium salt.Nomenclature
Sodium deoxycholate.Properties
Soly in water at 15°: >333 g/l.Therapeutic Category
Choleretic.
Keywords
Choleretic