2900. Deoxycorticosterone
Nomenclature
CAS number: 64-85-7
21-Hydroxypregn-4-ene-3,20-dione; 4-pregnen-21-ol-3,20-dione; 21-hydroxyprogesterone; desoxycorticosterone; 11-deoxycorticosterone; cortexone; desoxycortone; Kendall's desoxy compound B; Reichstein's substance Q. C21H30O3; mol wt 330.46.
C 76.33%, H 9.15%, O 14.52%.
Description and references
Occurs in adrenal cortex: Reichstein, von Euw, Helv. Chim. Acta 21, 1197 (1938); Steiger, Reichstein, ibid. 20, 1164 (1937). Numerous prepns from other steroids: Schindler et al., ibid. 24, 371 (1941); Reichstein, DE 875353 (1953 to Schering); Bockmühl et al., DE 871153 (1953 to Hoechst); Wettstein et al., US 2778776 (1957 to Ciba); NL 89575 (1958 to Organon); Kaspar et al., DE 1028572 (1958 to Schering). Isoln from the prothoracal glands of the water beetle, Dytiscus marginalis: Schildknecht et al., Angew. Chem. 78, 392 (1966).
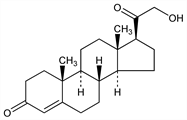
Properties
Plates from ether, mp 141-142°. [α]D22 +178° (alc). uv max: 240 nm. Freely sol in alcohol, acetone.Derivative
Acetate see Deoxycortiscosterone Acetate.Derivative
Tetraacetyl-β-d-glucoside.C35H48O12; mol wt 660.75.
C 63.62%, H 7.32%, O 29.06%.
Properties
Clusters of needles from 50% alc, mp 176-176.5°. [α]D24 +80° (c = 0.515 in chloroform). Believed to occur in nature in this glycosidic form: Johnson, J. Am. Chem. Soc. 63, 3238 (1941).Therapeutic Category
Mineralocorticoid.
Keywords
Mineralocorticoid