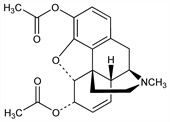
2968. Diacetylmorphine
Nomenclature
Description and references
Semisynthetic opioid analgesic. Prepn from morphine and acetic anhydride: O. Hesse, Ann. 220, 203 (1883); from morphine and acetyl chloride: Small, Lutz, Chemistry of the Opium Alkaloids, Supplement No. 103, Public Health Reports, Washington (1932); K. W. Bentley, The Chemistry of the Morphine Alkaloids (Oxford, 1954). Vapor pressure studies: A. H. Lawrence et al., Can. J. Chem. 42, 1886 (1984). Pharmacology: N. B. Eddy, H. A. Howes, J. Pharmacol. Exp. Ther. 53, 430 (1935). Pharmacodynamics: J. G. Umans, C. E. Inturrisi, Eur. J. Pharmacol. 85, 317 (1982). Clinical pharmacokinetics: F. Girardin et al., Clin. Pharmacol. Ther. 74, 341 (2003). Comprehensive description: D. K. Wyatt, L. T. Grady, Anal. Profiles Drug Subs. 10, 357-403 (1981). Review of determn methods in biological fluids: S. Pichini et al., Mass Spectrom. Rev. 18, 119-130 (1999). Use as adjunct to anesthesia for Caesarean section: S. Lane et al., Anaesthesia 60, 453 (2005). Review of use in maintenance treatment of heroin addiction: M. Ferri et al., J. Subst. Abuse Treat. 30, 63-72 (2006).
Properties
Orthorhombic plates, tablets from ethyl acetate. mp 173°. bp12 272-274°. [α]D25 -166° (c = 1.49 in methanol). One gram dissolves in 1.5 ml chloroform, 31 ml alcohol, 100 ml ether, 1700 ml water. Slightly sol in ammonia or sodium carbonate soln, sol in alkalies, dec by boiling with water. Turns pink and emits acetic odor on prolonged exposure to air. LD50 i.v. in mice: 59 μmol/kg (Umans, Inturrisi).Derivative
Hydrochloride monohydrate.Properties
Fine crystals, mp 243-244°. [α]D24 -156° (c = 1.044). Sol in 2 parts water, 11 parts alcohol. Insol in ether.Derivative
Methyl iodide.Properties
Needles, mp 252°, [α]D15 -107° (c = 0.896).Note
“China White” has been used as a term for very pure Southeast Asian heroin. This term has also been erroneously used to refer to 3-methylfentanyl and α-methylfentanyl, q.v., which are potent derivs of fentanyl, q.v. See: S. Stinson, Chem. Eng. News 59, 71 (Jan. 19, 1981).Note
This is a controlled substance (opium derivative): 21 CFR, 1308.11.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Phenanthrenes