3098. Didanosine
Nomenclature
Description and references
Purine nucleoside reverse transcriptase inhibitor (NRTI); converted in vivo to the active metabolite, dideoxyadenosine 5′-triphosphate, q.v. Enzymatic prepn from dideoxyadenosine: W. Plunkett, S. S. Cohen, Cancer Res. 35, 1547 (1975). Synthesis: R. R. Webb et al., Nucleosides Nucleotides 7, 147 (1988). Antiviral activity vs HIV-1: H. Mitsuya, S. Broder, Proc. Natl. Acad. Sci. USA 83, 1911 (1986). Antiretroviral spectrum in vitro: J. E. Dahlberg et al., ibid. 84, 2469 (1987). HPLC determn in human plasma: G. Ray, E. Murrill, Anal. Lett. 20, 1815 (1987). Comprehensive description: M. N. Nassar et al., Anal. Profiles Drug Subs. Excip. 22, 185-227 (1993). Review of pharmacokinetics and clinical experience in HIV infection: C. M. Perry, S. Noble, Drugs 58, 1099-1135 (1999); S. Moreno et al., ibid. 67, 1441-1462 (2007).
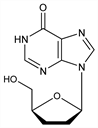
Properties
White crystalline powder. mp 160-163°. pKa 9.12 ±0.02. Partition coefficient (1-octanol/0.05M phosphate buffer pH 7): 0.068 ±0.005. [α]D25 -26.3° (c = 10 in water). uv max: 248 nm (pH 2); 254 nm (pH 12). Soly in water at 25° (mg/ml): 27.3 (pH 6); 429 (pH 10). Soly at 23° (mg/ml): <1 in acetone, acetonitrile, t-butanol, chloroform, ethanol, ethyl acetate, hexane, methylene chloride, 1-propanol, 2-propanol; 1 in PEG-300; 6 in methanol; 8 in propylene glycol; 45 in dimethylacetamide; 200 in DMSO. Unstable in acidic solns.Therapeutic Category
Antiretroviral.
Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides