3101. Dideoxyadenosine
Nomenclature
Description and references
Purine nucleoside reverse transciptase inhibitor (NRTI). Prepn: M. J. Robins, R. K. Robins, J. Am. Chem. Soc. 86, 3585 (1964); G. L. Tong et al., J. Org. Chem. 30, 2854 (1965); J. R. McCarthy et al., J. Am. Chem. Soc. 88, 1549 (1966). Chain-terminating inhibition of DNA synthesis: L. Toji, S. S. Cohen, Proc. Natl. Acad. Sci. USA 63, 871 (1969). Antiviral activity vs HIV-1: H. Mitsuya, S. Broder, ibid. 83, 1911 (1986). HPLC determn in biological fluids: P. A. Blau et al., J. Chromatogr. 420, 1 (1987).
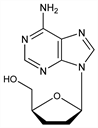
Properties
Crystals from ethanol, mp 184-186°. [α]D25 -25.2° (c = 1.01 in water). uv max in methanol: 259.5 nm (ε 14800).Derivative
5′-Triphosphate.Nomenclature
Description and references
Active metabolite of didanosine, q.v. RIA determn in blood cells: C. Le Saint et al., Antimicrob. Agents Chemother. 48, 589 (2004).
Properties
uv max: 261 nm (ε 15200).Use
Triphosphate in DNA sequencing as 3′-end chain terminator.Therapeutic Category
Antiretroviral.
Keywords
Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides; Reverse Transcriptase Inhibitor