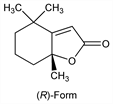
3170. Dihydroactinidiolide
Nomenclature
Description and references
Found in many plant sources including tea and tobacco. (-)-Form is naturally occurring. Produced by photo-oxidation of carotenoid flavor compounds. Pheromone component for queen recognition in red fire ants. Isoln from black tea aroma: J. Bricout et al., Helv. Chim. Acta 50, 1517 (1967); from Actinidia polygama Miq. and structure determn: T. Sakan et al., Tetrahedron Lett. 8, 1623 (1967); from tobacco and synthesis: W. C. Bailey, Jr. et al., J. Org. Chem. 33, 2819 (1968). Synthesis: S. Isoe et al., Tetrahedron Lett. 9, 5561 (1968); by photo-oxidation of β-carotene: eidem, ibid. 10, 279 (1969). Abs config study: eidem, ibid. 13, 2517 (1972). Prepn of enantiomers: K. Mori, Y. Nakazono, Tetrahedron 42, 283 (1986). Improved prepns: G. V. Subbaraju et al., Tetrahedron Lett. 32, 4871 (1991); A. Bosser et al., Biotechnol. Prog. 11, 689 (1995). Prepn of (R)-form: S. Yao et al., J. Org. Chem. 63, 118 (1998). Germination inhibition in wheat grains: T. Kato et al., J. Agric. Food Chem. 51, 2161 (2003).