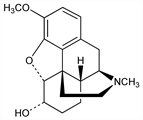
3171. Dihydrocodeine
Nomenclature
Description and references
Semisynthetic opioid analgesic; metabolized to dihydromorphine, q.v., by cytochrome P450 2D6. Prepd by reduction of codeine or neopine: Skita, Franck, Ber. 44, 2862 (1911); Wieland, Koralek, Ann. 433, 269 (1923); Stein, Pharmazie 10, 180 (1955). GC-MS determn in urine: M. Balikova et al., J. Chromatogr. B 752, 179 (2001). Metabolism in humans: M. F. Fromm et al., Clin. Pharmacol. Ther. 58, 374 (1995). Clinical pharmacokinetics: S. Ammon et al., Br. J. Clin. Pharmacol. 48, 317 (1999). Opioid receptor binding profile: H. Schmidt et al., Pharmacol. Toxicol. 91, 57 (2002). Clinical trial in chronic pain: C. H. Wilder-Smith et al., Pain 91, 23 (2001); in maintenance treatment of opioid dependence: J. R. Robertson et al., Addiction 101, 1752 (2006).
Properties
Crystals from methanol + water, mp 112-113°; bp15 248°.Derivative
Tartrate.Nomenclature
Properties
Crystals from methanol, contains 66.8% dihydrocodeine when completely anhydr. mp 192-193° (Stein). [α]D25 -72 to -75° (c = 1 in H2O). One gram dissolves in 4.5 ml water. Sparingly sol in alcohol. Insol in ether.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic; antitussive.
Keywords
Antitussive; Analgesic; Opioids; Phenanthrenes