3177. Dihydromorphine
Nomenclature
CAS number: 509-60-4
(5α,6α)-4,5-Epoxy-17-methylmorphinan-3,6-diol. C17H21NO3; mol wt 287.35.
C 71.06%, H 7.37%, N 4.87%, O 16.70%.
Description and references
Opioid analgesic; active metabolite of dihydrocodeine, q.v., and precursor in the manuf of hydromorphone, q.v. Prepd by hydrogenation of morphine: L. Oldenberg, Ber. 44, 1829 (1911); N. B. Eddy, J. G. Reid J. Pharmacol. 52, 468 (1934). Prepn from neopine: L. Small, J. Org. Chem. 12, 359 (1947). High yield synthesis from tetrahydrothebaine: A. K. Przybyl et al., ibid. 68, 2010 (2003). Comparative pharmacology: A.-K. Gilbert et al., Eur. J. Pharmacol. 492, 123 (2004).
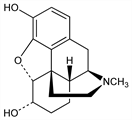
Derivative
Monohydrate.Properties
White crystals from alcohol, mp 155-157°. Insoluble in water. Sol in acetone, alcohol, chloroform, dil acids.Derivative
Hydrochloride.Nomenclature
CAS number: 1421-28-9
Paramorphan; paramorfan. C17H21NO3.HCl; mol wt 323.81.
C 63.06%, H 6.85%, N 4.33%, O 14.82%, Cl 10.95%.