3202. Diltiazem
Nomenclature
Description and references
Calcium channel blocker with vasodilating activity. Prepn (unspec stereochem): H. Kugita et al., DE 1805714; eidem, US 3562257 (1969, 1971 both to Tanabe Seiyaku); eidem, Chem. Pharm. Bull. 19, 595 (1971). Resolution of optical isomers: H. Inoue et al., Yakugaku Zasshi 93, 729 (1973), C.A. 79, 66331w (1974). Stereospecific synthesis: K. Igarashi, T. Honma, DE 3415035; eidem, US 4552695 (1984, 1985 both to Shionogi). Structure-activity studies: Sato et al., Arzneim.-Forsch. 21, 1338 (1971); T. Nagao et al., Chem. Pharm. Bull. 21, 92 (1973). Pharmacology and toxicity: eidem, Jpn. J. Pharmacol. 22, 467 (1972). Metabolism: Meshi et al., Chem. Pharm. Bull. 19, 1546 (1971). Review of synthesis and pharmacology: H. Inoue, T. Nagao, Chron. Drug Discovery 3, 207-238 (1993). Comprehensive description: D. J. Mazzo et al., Anal. Profiles Drug Subs. Excip. 23, 53-98 (1994). Review of pharmacology and efficacy in angina: M. Chaffman, R. N. Brogden, Drugs 29, 387-454 (1985); in hypertension: M. R. Weir, J. Clin. Pharmacol. 35, 220-232 (1995). Comparative clinical trial in prevention of complications of hypertension: L. Hansson et al., Lancet 356, 359 (2000).
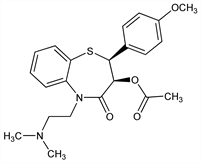
Derivative
Hydrochloride.Nomenclature
Properties
Fine needles from ethanol-isopropanol, mp 207.5-212°. Odorless, with a bitter taste. [α]D24 +98.3 ± 1.4° (c = 1.002 in methanol). Freely sol in water, methanol, chloroform, formic acid; slightly sol in abs ethanol. Practically insol in benzene. Insol in ether. LD50 in male, female mice, male, female rats (mg/kg): 61, 58, 38, 39 i.v.; 260, 280, 520, 550 s.c.; 740, 640, 560, 610 orally (Nagao, 1972).Therapeutic Category
Antianginal; antihypertensive; antiarrhythmic (class IV).
Therapeutic Category (Veterinary)
Antianginal; antihypertensive; antiarrhythmic (class IV).Keywords
Antianginal; Calcium Channel Blocker; Benzothiazepines