3296. Diosmetin
Nomenclature
CAS number: 520-34-3
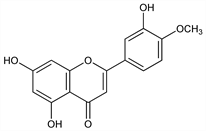
5,7-Dihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-1-benzopyran-4-one; 3′,5,7-trihydroxy-4′-methoxyflavone; cyanidenon-4′-methyl ether 1479; luteolin-4′-methyl ether. C16H12O6; mol wt 300.26.
C 64.00%, H 4.03%, O 31.97%.
Description and references
Aglycone of diosmin, q.v. Prepn from diosmin isolated from various plant sources: O. A. Oesterle, G. Wander, Helv. Chim. Acta 8, 519 (1925); isoln from lemons (Citrus limon Linn., Rutaceae): R. M. Horowitz, J. Org. Chem. 21, 1184 (1956). Synthesis and structural elucidation: G. Zemplén, R. Bognár, Ber. 76, 452 (1943). Synthesis: A. Lovecy et al., J. Chem. Soc. 1930, 817; N. B. Lorette et al., J. Org. Chem. 16, 930 (1951); J. H. Looker, M. J. Holm, ibid. 24, 1019 (1959). HPLC determn in biological fluids: D. Baylocq et al., Ann. Pharm. Fr. 41, 115 (1983).
Derivative
Hemimethanolate.C16H12O6. 1/2 CH3OH; mol wt 316.28.
C 62.66%, H 4.46%, O 32.88%.
Properties
Yellow needles from alcohol/ethyl acetate, sinters at 248°. mp 253-254°. Also reported as small yellow needles from methanol, mp 258-259° (Horowitz). uv max: 345, 268, 253 nm (log ε 4.32, 4.25, 4.28).Derivative
Triacetate.C22H18O9; mol wt 426.37.
C 61.97%, H 4.26%, O 33.77%.