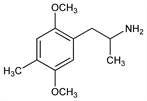
3412. DOM
Nomenclature
Description and references
Psychedelic compound of “hippie” culture. Prepn: A. T. Shulgin, US 3547999 (1970 to Dow). Pharmacology: B. T. Ho et al., J. Med. Chem. 13, 26 (1970). Clinical pharmacology: S. H. Snyder et al., Am. J. Psychiatry 125, 357 (1968). Mechanism of action: R. A. Glennon, Neuropsychopharmacology 3, 509 (1990); C. S. Aulakh et al., J. Pharmacol. Exp. Ther. 271, 143 (1994). Abuse potential: P. B. Silverman, B. T. Ho, Psychopharmacology 68, 209 (1980); D. Fiorella et al., ibid. 119, 239 (1995). Activity comparison of enantiomers: F. Benington et al., Nature 242, 185 (1973); sepn and determn of enantiomers by HPLC: J. Goto et al., J. Liq. Chromatogr. 2, 1179 (1979). Analysis by TLC: I. Ojanper"a et al., J. Planar Chromatogr. Mod. TLC 4, 373 (1991). Brief description including prepn: A. Shulgin, A. Shulgin, Pihkal (Transform Press, Berkeley CA, 1991) pp 637-642.