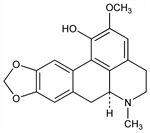
3413. Domesticine
Nomenclature
CAS number: 476-71-1
(6aS)-5,6,6a,7-Tetrahydro-2-methoxy-6-methyl-4H-benzo[de][1,3]benzodioxolo[5,6-g]quinolin-1-ol; 2-methoxy-9,10-(methylenedioxy)-6aα-aporphin-1-ol; 1-hydroxy-2-methoxy-9,10-methylenedioxyaporphine. C19H19NO4; mol wt 325.36.
C 70.14%, H 5.89%, N 4.30%, O 19.67%.
Description and references
In Nandina domestica Thunb., Berberidaceae. Isoln: Kitasato, Shishido, Ann. 527, 176 (1937). Syntheses: Govindachari et al., Indian J. Chem. 7, 841 (1969); Kessar et al., ibid. 8, 468 (1970); Kametani et al., J. Chem. Soc. C 1971, 2446, 2712; eidem, Chem. Pharm. Bull. 21, 766 (1973); Horii et al., ibid. 22, 583 (1974); Hoshino et al., ibid. 23, 2048 (1975).