3561. Emodin
Nomenclature
Description and references
Occurs mostly as the rhamnoside (see Frangulin) in rhubarb root, in alder buckthorn (Rhamnus frangula L.), in Cascara sagrada (Rhamnus purshiana DC., Rhamnaceae), also in Rumex and in other Polygonaceae. Isoln from rhubarb root: Tutin, Clewer, J. Chem. Soc. 99, 946 (1911); Carelli, Giuliano, Farmaco Ed. Prat. 12, 184 (1957); from bark of alder buckthorn: Bridel, Charaux, Bull. Soc. Chim. Biol. 15, 648 (1933). Identity with archin: Chaudhry et al., J. Sci. Ind. Res. 9B, No. 6, 142 (1950), C.A. 44, 9396h (1950). Synthesis from 3,5-dinitrophthalic anhydride and m-cresol: Elder, Widmer, Helv. Chim. Acta 6, 966 (1923); from 2-methylanthraquinone: Ayyangar et al., J. Sci. Ind. Res. 20B, 493 (1961), C.A. 57, 8514b (1962).
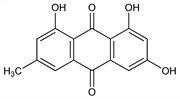
Properties
Orange needles from alc or by sublimation at 12 mm. mp 256-257°. Absorption max (ethanol): 222, 252, 265, 289, 437 nm (log ε 4.55, 4.26, 4.27, 4.34, 4.10). Practically insol in water; sol in alc, aq alkali hydroxide solns (cherry-red color), Na2CO3 and NH3 solns. Soly at 25° (g/100 ml of satd soln): ether 0.140; chloroform 0.071; carbon tetrachloride 0.010; carbon bisulfide 0.009; benzene 0.041.Derivative
3-Methyl ether.Nomenclature
1,8-Dihydroxy-3-methoxy-6-methylanthraquinone; rheochrysidin.Properties
Brick-red, monoclinic needles, mp 207°. Occurs naturally as physcione or parietin.Derivative
Trimethyl ether.Properties
Pale yellow needles, mp 225°.Therapeutic Category
Cathartic.
Keywords
Laxative/Cathartic