3565. Emtricitabine
Nomenclature
Description and references
Reverse transcriptase inhibitor; nucleoside analog structurally related to lamivudine, q.v. Prepn: D. C. Liotta et al., WO 9214743 (1992 to Emory University); G. Dionne, US 5538975 (1996 to BioChem Pharma, Inc.); L. S. Jeong et al., J. Med. Chem. 36, 181 (1993). Absolute configuration: P. van Roey et al., Antivir. Chem. Chemother. 4, 369 (1993). HPLC-NMR determn of urinary metabolites: J. P. Shockcor et al., Xenobiotica 26, 189 (1996). Comparative efficacy of enantiomers vs HIV: R. F. Schinazi et al., Antimicrob. Agents Chemother. 36, 2423 (1992). Pharmacokinetics: L. W. Frick et al., ibid. 38, 2722 (1994). Mechanism of action study: J. Y. Feng et al., FASEB J. 13, 1511 (1999). Clinical study in combination with didanosine and efavirenz: J.-M. Molina et al., J. Infect. Dis. 182, 599 (2000). Review: P. Cahn, Expert Opin. Invest. Drugs 13, 55-68 (2004).
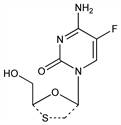
Properties
White solid from ether and methanol, mp 136-140°. [α]D25 -133.60° (c = 0.23 in MeOH). uv max (water): 287.8 nm (pH 2); 280.0 nm (pH 7); 279.8 nm (pH 11) (ε 14210, 11090, 11810).Therapeutic Category
Antiretroviral.
Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides