3590. Enoxolone
Nomenclature
CAS number: 471-53-4
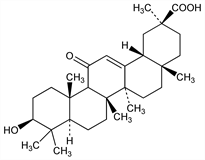
(3β,20β)-3-Hydroxy-11-oxoolean-12-en-29-oic acid; 3β-hydroxy-11-oxoolean-12-en-30-oic acid; glycyrrhetic acid; 18β-glycyrrhetinic acid; uralenic acid; Arthrodont (Veyron-Froment); Biosone (Berk); P.O. 12 (Biothérax). C30H46O4; mol wt 470.68.
C 76.55%, H 9.85%, O 13.60%.
Description and references
From glycyrrhizic acid, q.v. Structure: Ruzicka et al., Helv. Chim. Acta 26, 2143, 2278 (1943). Stereochemistry: Beaton, Spring, J. Chem. Soc. 1955, 3126. Prepn from shredded licorice root: Mer, Am. Perfum. Aromat. 74(6), 39 (1959). Manuf: GB 833184 (1960 to Carlo Erba). Identity with uralenic acid: Belous et al., Zh. Obshch. Khim. 35, 401 (1965). Metabolism: Parke et al., J. Pharm. Pharmacol. 15, 500 (1963). Mechanism of action: Helbing, Berntsen, Pharm. Weekbl. 100, 1438 (1965).
Properties
Needles from alcohol + petr ether, mp 296°. [α]D21 +86° (alc); [α]D20 +145.5° (dioxane); [α]D20 +163° (chloroform). Freely sol in chloroform, dioxane. Soluble in alcohol, pyridine, acetic acid. Practically insol in petr ether.Derivative
18α-Hydrogen Form.Properties
Platelets from dil alcohol, mp 335°. [α]D20 +140° (alcohol); [α]D +98° (c = 0.1 in chloroform). Sol in alcohol, dioxane, chloroform.Therapeutic Category
Anti-inflammatory (topical).
Keywords
Glucocorticoid