4505. Glycyrrhizic Acid
Nomenclature
Description and references
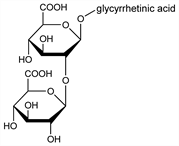
Triterpene saponin used in traditional Chinese medicinal preparations for its anti-inflammatory, antiulcerous and antiallergic effects. Extraction from Glycyrrhiza glabra L., Leguminosae: Karrer, Chao, Helv. Chim. Acta 4, 100 (1921); Ruzicka, Louenberger, ibid. 19, 1402 (1936). From commercial glycyrrhizinum ammoniacale: Tschirch, Cederberg, Arch. Pharm. 245, 97 (1907); Voss et al., Ber. 70, 122 (1937). Revised method of isoln: Conn, Conn, J. Lab. Clin. Med. 47, 20 (1956). Structure: Lythgoe, Trippett, J. Chem. Soc. 1950, 1983. Revised structure: Marsh, Levvy, Biochem. J. 63, 9 (1956). See also: I. Kitagawa et al., Chem. Pharm. Bull. 36, 3710 (1988); T. Hatano et al., ibid. 39, 1238 (1991). Synthesis of derivatives: Brieskorn, Sax, Arch. Pharm. 303, 905 (1970). LC-MS/MS determn in plasma: Z. J. Lin et al., J. Chromatogr. B 814, 201 (2005). Review: Nieman, Chem. Weekbl. 48, 213 (1952).