3648. Ergocornine
Nomenclature
CAS number: 564-36-3
(5′α)-12′-Hydroxy-2′,5′-bis(1-methylethyl)ergotaman-3′,6′,18-trione. C31H39N5O5; mol wt 561.67.
C 66.29%, H 7.00%, N 12.47%, O 14.24%.
Description and references
Natural ergot alkaloid derived from lysergic acid; component of ergotoxine, q.v. Isoln from ergot: Stoll, Hofmann, Helv. Chim. Acta 26, 1570 (1943). Structure: Stoll et al., ibid. 34, 1544 (1951). Separation and purification: Stoll, Hofmann, US 2447214 (1948 to Sandoz). Synthesis: Stadler et al., Helv. Chim. Acta 52, 1549 (1969). Determn in ergot by capillary electrophoresis: K. Frach, G. Blaschke, J. Chromatogr. A 808, 247 (1998).
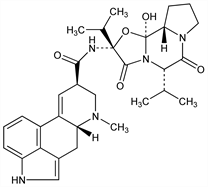
Properties
Solvated, polyhedra from methanol, dec 181° (contains 1 mole methanol). [α]D20 -110° (pyridine); -175° (chloroform). uv max (methanol): 311 nm (log ε 3.91). Soluble in acetone, chloroform, ethyl acetate; slightly sol in ethyl and methyl alcohol. Nearly insol in water.Derivative
8α-Epimer.Nomenclature
CAS number: 564-37-4
Ergocorninine.