3659. Ergosterol
Nomenclature
Description and references
Most important of the provitamins D. Usually obtained from yeast which synthesizes it from simple sugars such as glucose. Damp yeast yields about 2.5% ergosterol, the variety of the yeast being very important. Isoln procedure: Green et al., US 3006932 (1961 to Vitamins Ltd.). When irradiated with uv light, ergosterol develops powerful vitamin D2, q.v., activity. Askew et al., in England and Windaus and collaborators in Germany isolated the antirachitic vitamin D2. The main irradiation products of ergsterol are lumisterol→tachysterol→vitamin D2. Structure: Chuang, Ann. 500, 270 (1933). Oxidation products: Fuerst, Arch. Pharm. 300, 144 (1967).
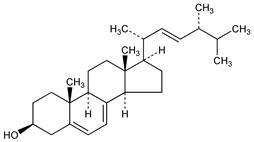
Properties
Small hydrated plates from alcohol, in hydrated needles from ether. The best crystallized form contains 1 1/2 mol H2O, mp 168°: Bills, Honeywell, J. Biol. Chem. 80, 15 (1928). Complete removal of water is almost impossible and results in an amorphous mass, melting range 166-183°. bp0.01 250°. [α]D20 -135° (c = 1.2 in CHCl3 calcd as anhydr). [α]20546 -171° (CHCl3). uv max (ethanol): 262, 271, 282, 293 nm, Hogness et al., ibid. 120, 239 (1937). Practically insol in water. One gram dissolves in 660 ml alcohol, in 45 ml boiling alcohol, in 70 ml ether, in 39 ml boiling ether, in 31 ml chloroform. Precipitated by digitonin. Affected by light and air, turns yellow. Oxygen forms peroxides and hydrogen may form polyhydro compds.Derivative
Acetate.Properties
mp 179°, clear at 181°.Derivative
Benzoate.Properties
Plates or needles, mp 169-171°. [α]D23 -71° (c = 1.1 in CHCl3), [α]546 -88° (CHCl3).Derivative
22,23-Dihydro analog.Nomenclature
22,23-Dihydroergosterol.Description and references
Prepn: A. Windhaus, R. Langer, Ann. 508, 105 (1934); D. H. R. Barton et al., J. Chem. Soc. Perkin Trans. 1 1976, 821; D. J. Curry et al., ibid. 1977, 822. uv irradiation gives vitamin D4, q.v.
Properties
Solvated needles from ethyl acetate + methanol, mp 152-153° (dried). [α]D19 -109° (CHCl3) (Windhaus); also reported as needles from chloroform-methanol, mp 128-130°. [α]D19 -121° (c = 0.1). uv max: 262, 272, 282, 294 nm (ε 8000, 11200, 11800, 6800) (Barton).Derivative
22,23-Dihydro analog acetate.Properties
mp 157-158°. [α]D17 -75° (c = 2.1 in CHCl3).Therapeutic Category
Vitamin (antirachitic).
Keywords
Vitamin/Vitamin Source; Vitamin D