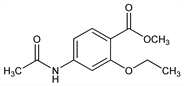
3745. Ethopabate
Nomenclature
CAS number: 59-06-3
4-Acetamido-2-ethoxybenzoic acid methyl ester; methyl 4-acetamido-2-ethoxybenzoate; 2-ethoxy-4-acetamidobenzoic acid methyl ester; ethyl pabate. C12H15NO4; mol wt 237.25.
C 60.75%, H 6.37%, N 5.90%, O 26.97%.
Description and references
Prepn: Grimme, Schmitz, Ber. 87, 179 (1954); E. F. Rogers, R. L. Clark, BE 613166; eidem, US 3211610 (1962, 1965 both to Merck & Co.); FR 1407055; M. L. Thominet, US 3357978 (1965, 1967 both to Soc. d'Etudes Sci. Ind. de l'Ile-de-France). Anticoccidial activity studies: Rogers et al., Proc. Soc. Exp. Biol. Med. 117, 488 (1964). Metabolism in chickens: Buhs et al., J. Pharmacol. Exp. Ther. 154, 357 (1966).