3747. Ethopropazine
Nomenclature
CAS number: 522-00-9
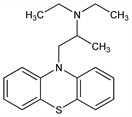
N,N-Diethyl-α-methyl-10H-phenothiazine-10-ethanamine; 10-(2-diethylaminopropyl)phenothiazine; 10-(2-diethylamino-2-methylethyl)phenothiazine; 2-diethylamino-1-propyl-N-dibenzoparathiazine; phenopropazine; profenamine; RP-3356; W-483; Isothazine; Isothiazine; Parkin (Yoshitomi). C19H24N2S; mol wt 312.47.
C 73.03%, H 7.74%, N 8.97%, S 10.26%.
Description and references
Anticholinergic. Prepd from Grignard complexes of diethylaminopropyl halide and phenothiazine: S. S. Berg, J. N. Ashley, US 2607773 (1952 to Rh^one-Poulenc). Pharmacology: M. E. Farquharson, R. G. Johnston, Br. J. Pharmacol. 14, 559 (1959). Pharmacokinetics: M. Maboudian-Esfahani, D. R. Brocks, Biopharm. Drug Dispos. 20, 159 (1999). HPLC determn in plasma: eidem, J. Chromatogr. B 715, 417 (1998).
Properties
Crystals, mp 53-55°. Usually obtained as an oil because of contamination with 10-(2-diethylamino-1-methylethyl)phenothiazine.Derivative
Hydrochloride.Nomenclature
CAS number: 1094-08-2
Dibutil (Bayer); Lysivane (M & B); Pardisol; Parphezein; Parphezin; Parsidol (Warner-Chilcott; Specia); Parsitan; Parsotil; Rodipal. C19H24N2S.HCl; mol wt 348.93.
C 65.40%, H 7.22%, N 8.03%, S 9.19%, Cl 10.16%.
Properties
Crystals from ethylene dichloride, mp 223-225° (some decompn). Lower melting points reported are caused by admixture with 10-(2-diethylamino-1-methylethyl)phenothiazine-HCl which melts at 166-168°. One gram dissolves in 400 ml water at 20°, in 20 ml water at 40°. Sol in ethanol, chloroform. Soly in abs ethanol at 25° = 1.0 g/30 ml. Sparingly sol in acetone. Practically insol in ether, benzene. pH of a 5% aq soln is about 5.8. LD50 s.c. in mice: 670 mg/kg (Farquharson, Johnston).Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian; Antimuscarinic