3881. Etomidate
Nomenclature
Description and references
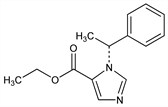
Imidazole hypnotic used in the induction of anesthesia. Prepn of the (±)-form: BE 662474; E. F. Godefroi, C. A. M. van der Eijcken, US 3354173 (1965, 1967 both to Janssen); E. F. Godefroi et al., J. Med. Chem. 8, 220 (1965). Prepn of (R)-(+)-form: L. F. C. Roevens et al., DE 2609573; eidem, US 3991072 (both 1976 to Janssen). Metabolism: E. Goetz, Anaesthesist 23, 331 (1974). Pharmacokinetics: M. J. Van Hamme et al., Anesthesiology 49, 274 (1978). Pharmacology: P. A. J. Janssen et al., Arch. Int. Pharmacodyn. Ther. 214, 92 (1975). HPLC determn in plasma: M. P. McIntosh, R. A. Rajewski, J. Pharm. Biomed. Anal. 24, 689 (2001). Clinical study: R. J. Fragen, N. Caldwell, Anesthesiology 50, 242 (1979). Clinical trial vs. midazolam in pediatric sedation and analgesia: L. Di Liddo et al., Ann. Emerg. Med. 48, 433 (2006). Comprehensive description: Z. L. Chang, J. B. Martin, Anal. Profiles Drug Subs. 12, 191-214 (1983). Veterinary use: S. Robertson, Vet. Clin. North Am. Small Anim. Pract. 22, 277 (1992). Review of use in rapid sequence intubation: A. J. Oglesby, Emerg. Med. J. 21, 655-659 (2004).
Properties
Crystals from diisopropyl ether, mp 67°. [α]D20 +66° (c = 1 in ethanol). uv max (isopropanol): 240 nm (ε 12200). Soly in water at 25°: 0.0045 mg/100 ml. Sol in chloroform, methanol, ethanol, propylene glycol, acetone. LD50 in mice, rats (mg/kg): 29.5, 14.8-24.3 i.v. (Janssen).Therapeutic Category
Anesthetic (intravenous).
Therapeutic Category (Veterinary)
Anesthetic (intravenous).Keywords
Sedative/Hypnotic