4066. Fervenulin
Nomenclature
CAS number: 483-57-8
6,8-Dimethylpyrimido[5,4-e]-1,2,4-triazine-5,7-(6H,8H)-dione; 6,8-dimethyl-5,7-dioxo-5,6,7,8-tetrahydropyrimido[5,4-e]-as-triazine; 1,3-dimethylazalumazine; planomycin. C7H7N5O2; mol wt 193.16.
C 43.53%, H 3.65%, N 36.26%, O 16.57%.
Description and references
Antibiotic from culture filtrates of Streptomyces fervens: Eble et al., Antibiot. Annu. 1959-1960, 227. Structure: Daves et al., J. Org. Chem. 26, 5256 (1961). Synthesis: Pfleiderer, Schündehütte, Ann. 615, 42 (1958); Daves et al., J. Am. Chem. Soc. 84, 1724 (1962); Yoneda, Nagamatsu, Bull. Chem. Soc. Jpn. 48, 2884 (1975); Taylor, Sowinski, J. Org. Chem. 40, 2321 (1975); S. Senda et al., J. Am. Chem. Soc. 99, 7358 (1977).
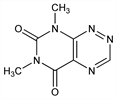
Properties
Yellow orthorhombic crystals, mp 178-179°. uv max (ethanol): 238, 275, 340 nm (ε 18,500, 1600, 4200). Sol in practically all of the common organic solvents; sol in cold water to about 2 mg/ml; in hot water to about 40 mg/ml. Practically insol in hydrocarbons. Labile to alkali; stable to acid.Derivative
4-Oxide.C7H7N5O3; mol wt 209.16.
C 40.20%, H 3.37%, N 33.48%, O 22.95%.
Description and references
Synthesis: M. Ichiba et al., J. Heterocycl. Chem. 14, 175 (1977); K. Senga et al., Heterocycles 6, 273 (1977); synthesis and conversion to fervenulin: M. Ichiba et al., J. Org. Chem. 43, 469 (1978).