4195. Flupyrsulfuron-methyl
Nomenclature
CAS number: 144740-53-4
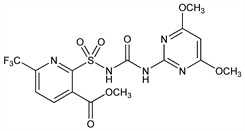
2-[[[[(4,6-Dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]-6-(trifluoromethyl)-3-pyridinecarboxylic
acid methyl ester; methyl 2-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-6-(trifluoromethyl)nicotinate. C15H14F3N5O7S; mol wt 465.36.
C 38.71%, H 3.03%, F 12.25%, N 15.05%, O 24.07%, S 6.89%.
Description and references
Sulfonylurea postemergent herbicide which inhibits acetolactate synthetase. Prepn: T. A. Andrea, P. H. T. Liang, EP 502740; eidem, US 5393734 (1992, 1995 both to Du Pont). Description of chemical and biological activities: S. R. Teaney et al., Brighton Crop Prot. Conf. - Weeds 1995, 49. Metabolism and selectivity: M. K. Koeppe et al., Pestic. Biochem. Physiol. 59, 105 (1998). LC determn in soil and water: C. R. Powley, P. A. de Bernard, J. Agric. Food Chem. 46, 514 (1998). Environmental fate: S. K. Singles et al., Pestic. Sci. 55, 288 (1999).
Properties
Solid, mp 130-137°.Derivative
Sodium salt.Nomenclature
CAS number: 144740-54-5
DPX-KE459; Lexus (DuPont); Millenium (DuPont). C15H13F3N5NaO7S; mol wt 487.34.
C 36.97%, H 2.69%, F 11.70%, N 14.37%, Na 4.72%, O 22.98%, S
6.58%.