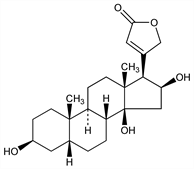
4433. Gitoxigenin
Nomenclature
CAS number: 545-26-6
(3β,5β,16β)-3,14,16-Trihydroxycard-20(22)-enolide; Δ20,22-3,14,16,21-tetrahydroxynorcholenic acid lactone. C23H34O5; mol wt 390.51.
C 70.74%, H 8.78%, O 20.49%.
Description and references
The aglycon of gitoxin. By refluxing gitoxin in a mixture of water + alcohol + HCl: Smith, J. Chem. Soc. 1931, 23. Structure: Jacobs, Elderfield, J. Biol. Chem. 100, 671 (1933); Elderfield, Chem. Rev. 17, 217 (1935); Henderson, Chen, J. Med. Pharm. Chem. 5, 988 (1962). Configuration: Moore, Helv. Chim. Acta 37, 659 (1954); Repke, Klesczewski, Arch. Exp. Pathol. Pharmakol. 239, 131 (1960). Cf. ref under Digoxigenin.