4481. Glyceraldehyde
Nomenclature
Description and references
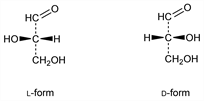
Obtained with its isomer dihydroxyacetone from glycerol by mild oxidation: Witzemann, J. Am. Chem. Soc. 36, 2227 (1914). See also Org. Synth. coll. vol. II, 305 (1943). The equilibrium mixture of glyceraldehyde and dihydroxyacetone is called glycerose. The two isomers are convertible into another through a common enediol resulting from the migration of hydrogen atoms (Lobry de Bruyn-van Eckenstein rearrangement). The equilibrium mixture plays an important role in the fermentation of sugars and in the biogenesis of constituents of the animal organism, cf. L.F. Fieser, M. Fieser, Advanced Organic Chemistry (Reinhold, New York, 1961) pp 78, 284, 405. Glyceraldehyde is the simplest aldose; the d- and l-forms are the configurational reference standard for carbohydrates. The two forms have been obtained through the action of nitrous acid on the corresponding form of 3-amino-2-hydroxypropanal: Wohl, Momber, Ber. 47, 3346 (1914); Pictet, Barbier, Helv. Chim. Acta 4, 924 (1921); cf. Baer, Fischer, Science 88, 108 (1938). Prepn of l-glyceraldehyde from l-sorbose and of d-glyceraldehyde from d-fructose: Perlin, Methods Carbohydr. Chem. 1, 61 (1962).