4591. Halcinonide
Nomenclature
CAS number: 3093-35-4
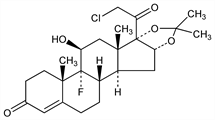
(11β,16α)-21-Chloro-9-fluoro-11-hydroxy-16,17-[(1-methylethylidene)bis(oxy)]pregn-4-ene-3,20-dione; 21-chloro-9-fluoro-11β,16α,17-trihydroxypregn-4-ene-3,20-dione
cyclic 16,17-acetal with acetone; 21-chloro-9α-fluoro-11β-hydroxy-16α,17α-isopropylidenedioxy-4-pregnene-3,20-dione; 9α-fluoro-21-chloro-11β,16α,17α-trihydroxypregn-4-ene-3,20-dione
16,17-acetonide; SQ-18566; Halciderm (BMS); Halcimat (BMS); Halog (BMS). C24H32ClFO5; mol wt 454.96.
C 63.36%, H 7.09%, Cl 7.79%, F 4.18%, O 17.58%.
Description and references
Prepn: Bernstein et al., J. Org. Chem. 27, 690 (1962); L. T. Difazio, M. A. Augustine, DE 2355710; eidem, US 3892857 (1972, 1975 both to Squibb). Pharmacological evaluation: Bagatell, Augustine, Curr. Ther. Res. 16, 748 (1974); R. C. Millonig, E. Yiakas, in Pharmacological and Biochemical Properties of Drug Substances vol. 1, M. E. Goldberg, Ed. (Am. Pharmaceut. Assoc., Washington, DC, 1977) pp 215-231. Comprehensive description: J. Kirshbaum, Anal. Profiles Drug Subs. 8, 251-281 (1979).
Properties
Crystals from acetone-petr ether, mp 264-265° (dec). uv max (methanol): 238 nm (ε 16400). [α]D25 +155° (CHCl3). Sol in acetone, chloroform, DMSO. Slightly sol in benzene, ethanol, ethyl ether, methanol. Insol in water, 0.1M HCl, 0.1M NaOH, hexanes. LD50 i.p. in mice: 150 mg/kg (Millonig, Yiakas).Therapeutic Category
Anti-inflammatory (topical).
Keywords
Glucocorticoid