4628. Helicin
Nomenclature
CAS number: 618-65-5
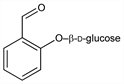
2-(β-d-Glucopyranosyloxy)benzaldehyde; salicylaldehyde β-d-glucoside. C13H16O7; mol wt 284.26.
C 54.93%, H 5.67%, O 39.40%.
Description and references
Prepn by the oxidation of salicin with dil nitric acid: Schiff, Ann. 154, 19 (1870); from salicylaldehyde + O-tetraacetyl-α-glucosidyl bromide: Robertson, Waters, J. Chem. Soc. 1930, 2729. ORD and stereochemical studies: Tsuzuki et al., Bull. Chem. Soc. Jpn. 44, 526 (1971).
Properties
Needles with 0.75 mol H2O from H2O; mp 175-176° when dried at 100°. [α]D20 60° (c = 1.4). One gram dissolves in 55 ml water; freely sol in hot water, alcohol. Forms compds with urea, thiourea, certain amino acids.Derivative
Tetraacetate.C21H24O11; mol wt 452.41.
C 55.75%, H 5.35%, O 38.90%.