4802. Hydrohydrastinine
Nomenclature
CAS number: 494-55-3
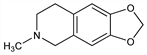
5,6,7,8-Tetrahydro-6-methyl-1,3,dioxolo[4,5-g]isoquinoline. C11H13NO2; mol wt 191.23.
C 69.09%, H 6.85%, N 7.32%, O 16.73%.
Description and references
Prepn from cotarnine, q.v.: Topchiev, J. Appl. Chem. USSR 6, 529 (1933), C.A. 28, 2718 (1934); Clayson, J. Chem. Soc. 1949, 2016; Schneider, Müller, Ann. 615, 34 (1958); Knabe, Arch. Pharm. 292, 652 (1959). See also: Hydrastinine, Hydrocotarnine.
Properties
Crystals from petr ether, mp 66°. bp752 303°. Sol in alc, ether, acetone, benzene, CS2, ethyl acetate. Absorption spectrum: Dobbie, Tinkler, J. Chem. Soc. 85, 1007 (1904).Derivative
Hydrochloride.Nomenclature
CAS number: 5985-04-6
C11H13NO2.HCl; mol wt 227.69.
C 58.03%, H 6.20%, N 6.15%, O 14.05%, Cl 15.57%.
Properties
Crystals from water or alcohol, dec 278°. Sol in water.Derivative
Hydrobromide.Nomenclature
CAS number: 5985-05-7
C11H13NO2.HBr; mol wt 272.14.
C 48.55%, H 5.19%, N 5.15%, O 11.76%, Br 29.36%.
Properties
Needles from water, dec 272°.Derivative
Hydriodide.C11H13NO2.HI; mol wt 319.14.
C 41.40%, H 4.42%, N 4.39%, O 10.03%, I 39.76%.
Properties
Crystals from water, dec 242°. Sol in water or alc.Derivative
Platinichloride.Nomenclature
CAS number: 5985-06-8
(C11H13NO2)2.H2PtCl6; mol wt 792.27.
C 33.35%, H 3.56%, N 3.54%, O 8.08%, Pt 24.62%, Cl 26.85%.