4820. 24-Hydroxycholesterol
Nomenclature
CAS number: 474-73-7
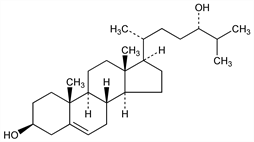
(3β,24S)-Cholest-5-ene-3,24-diol. C27H46O2; mol wt 402.65.
C 80.54%, H 11.52%, O 7.95%.
Description and references
Two stereoisomers exist: cholest-5-ene-3β,24β-diol and cholest-5-ene-3β,24α-diol. Isoln of the naturally occurring 24β-epimer, from horse brain: Ercoli et al., Boll. Soc. Ital. Biol. Sper. 29, 494 (1953), C.A. 49, 4744i (1955); from beef spinal cord: Fieser et al., J. Org. Chem. 22, 1380 (1957). Prepn and separation of isomers: Ercoli, Ruggieri, Gazz. Chim. Ital. 83, 720 (1953); eidem, J. Am. Chem. Soc. 75, 3284 (1953). Configurations at C-24: Klyne, Stokes, J. Chem. Soc. 1954, 1979.
Derivative
24β-Epimer.Nomenclature
Cerebrostenediol; cerebrosterol.Properties
Needles from acetone, mp 175-176°. [α]D20 48.2° (c = 1.06 in CHCl3).Derivative
24β-Epimer dibenzoate.C41H54O4; mol wt 610.87.
C 80.61%, H 8.91%, O 10.48%.