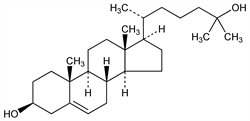
4821. 25-Hydroxycholesterol
Nomenclature
Description and references
An important intermediate in the synthesis of 25-hydroxycholecalciferol, q.v. Synthesis: A. Ryer et al., J. Am. Chem. Soc. 72, 4247 (1950); W. G. Dauben, H. L. Bradlow, ibid. 4248. Isoln from autoxidation products of cholesterol, q.v.: L. F. Fieser et al., J. Org. Chem. 22, 1380 (1957). Prepn from pregnenolone, q.v.: T. A. Narwid, M. R. Uskokovic, US 3856780 (1974). Alternate syntheses: M. Morisaki et al., Chem. Pharm. Bull. 21, 457 (1973); A. Rotman, Y. Mazur, Chem. Commun. 1974, 15; J. J. Partridge et al., Helv. Chim. Acta 57, 764 (1974); T. A. Narwid et al., ibid. 771; W. G. Salmond et al., Tetrahedron Lett. 1977, 987, 1237, 1695; K. Ochi et al., Chem. Pharm. Bull. 27, 252 (1979); M. Riediker, J. Schwartz, Tetrahedron Lett. 22, 4655 (1981).