4847. 5-Hydroxytryptophan
Nomenclature
Description and references
Precursor of serotonin. Synthesis from 5-benzyloxyindole: Ek, Witkop, J. Am. Chem. Soc. 76, 5579 (1954); Shaw, Morris, Biochem. Prep. 9, 92 (1962); from 5-benzyloxytryptophan: Frangatos, Chubb, Can. J. Chem. 37, 1374 (1959); Frangatos, CA 619472 (1961 to Frank W. Horner); Ash, GB 845034 (1960 to May & Baker); from tryptophan: Renson et al., Biochem. Biophys. Res. Commun. 6, 20 (1961). Prepn of l- and d-forms: A. J. Morris, M. D. Armstrong, J. Org. Chem. 22, 306 (1957). Crystal and molecular structure of dl-form: Wakahara et al., Tetrahedron Lett. 1970, 3003. Use of l-5HTP in treatment of myoclonus: M. H. Van Woert, D. Rosenbaum, Adv. Neurol. 26, 107 (1979). Clinical trial in migraine: F. Titus et al., Eur. Neurol. 25, 327 (1986). Review: M. H. Van Woert, Orphan Drugs, F. E. Karch, Ed. (Marcel Dekker, New York, 1982) pp 13-31. Review of antidepressant efficacy: W. F. Byerley et al., J. Clin. Psychopharmacol. 7, 127-137 (1987); of metabolism and clinical experience: T. C. Birdsall, Altern. Med. Rev. 3, 271-280 (1998).
Derivative
dl-Form.Properties
Minute rods or needles from ethanol, dec 298-300°. uv max (H2O at pH 6.0): 278 nm. Soly in water at 5°: 1.0 g/100 ml; at 100°: 5.5 g/100 ml. Soly in 50% boiling alc: 2.5 g/100 ml. Aq solns are stable at low pH.Derivative
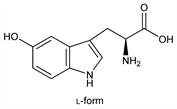
l-Form.Nomenclature
Properties
Crystals, [α]D20 32.5° (H2O); [α]D20 +16.0° (4N HCl).Derivative
d-Form.Nomenclature
Properties
Crystals, [α]D20 +32.2° (H2O).Therapeutic Category
Antidepressant; in treatment of myoclonus.
Keywords
Oxitriptan: Antidepressant; Bicyclics