10285. Hydroxytyrosol
Nomenclature
Description and references
Powerful phenolic antioxidant found in olive oil and other foods; exists in olive oil primarily in esterified form with elenolic acid as oleuropein aglycone. Prepn: C. Schpf et al., Ann. 563, 86 (1949); P. G. Baraldi et al., ibid. 1983, 684. Synthesis and recovery from olive oil mill waste waters: R. Capasso et al., J. Agric. Food Chem. 47, 1745 (1999) DOI PubMed. Photochemical synthesis from tyrosol, q.v.: S. Azabou et al., ibid. 55, 4877 (2007) DOI PubMed. GC/MS determn in wine: D. Di Tommaso et al., J. High Resolut. Chromatogr. 21, 549 (1998) DOI. Metabolism study using capillary GC/MS determn in plasma and urine: E. Miro-Casas et al., Clin. Chem. 49, 945 (2003) DOI PubMed. Antioxidant activity: M. De Lucia et al., Tetrahedron 62, 1273 (2006) DOI; S. J. Rietjens et al., J. Agric. Food Chem. 55, 7609 (2007) DOI PubMed.
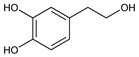
Properties
Colorless oil. uv max (methanol): 218, 281 nm (ε 4300, 1970). Log P (octanol/water): 0.03 ±0.01 (pH 5.5); 0.02 ±0.01 (pH 7.4).Derivative
Acetate.Nomenclature
Description and references
Prepn and antioxidant activity: M. H. Gordon et al., J. Agric. Food Chem. 49, 2480 (2001) DOI PubMed.