5228. Isotretinoin
Nomenclature
Description and references
Naturally occurring metabolite of vitamin A, q.v.; inhibits sebum production. Prepn: C. D. Robeson et al., J. Am. Chem. Soc. 77, 4111 (1955). Stereoselective process: R. Lucci, EP 111325; idem, US 4556518 (1984, 1985 both to Hoffmann-La Roche). Toxicology and teratogenicity study: J. J. Kamm, J. Am. Acad. Dermatol. 6, 652 (1982). Identification as endogenous metabolite of all-trans-retinoic acid: M. E. Cullum, M. H. Zile, J. Biol. Chem. 260, 10590 (1985). HPLC determn in serum: G. Tang, R. M. Russell, J. Lipid Res. 31, 175 (1990). GC determn in pharmaceutical formulations: E. M. Lima et al., J. Pharm. Biomed. Anal. 38, 678 (2005). Review of pharmacology and clinical efficacy in acne: A. R. Shalita et al., Cutis 42, Suppl. 6A, 1-19 (1988). Symposium on clinical experience: Dermatology 195, Suppl. 1, 1-37 (1997). Review of safety profile: A. Charakida et al., Expert Opin. Drug Saf. 3, 119-129 (2004).
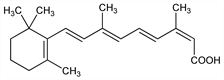
Properties
Reddish-orange plates from isopropyl alcohol, mp 174-175°. uv max: 354 nm (ε 39800). Practically insol in water. Sol in chloroform; sparingly sol in alcohol, isopropyl alcohol, polyethylene glycol 400. LD50 (20 day) in mice, rats (mg/kg): 904, 901 i.p.; 3389, >4000 orally (Kamm).Therapeutic Category
Antiacne.
Therapeutic Category (Veterinary)
In treatment of dermatologic conditions.Keywords
Antiacne