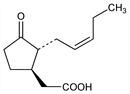
5260. Jasmonic Acid
Nomenclature
Description and references
Naturally occurring plant growth inhibitor. Orginally isolated from jasmin flowers as the methyl ester, the major aroma component: E. Demole et al., Helv. Chim. Acta 45, 675 (1962). Isoln of the free acid from Lasiodiplodia theobromae: D. C. Aldridge et al., J. Chem. Soc. 1971, 1623; by fermentation: D. Broadbent et al., GB 1286266 (1972 to ICI). Occurrence in plants: A. Meyer et al., J. Plant Growth Regul. 3, 1 (1984). NMR characterization: A. Husain et al., J. Nat. Prod. 56, 2008 (1993). Quantification by GC/MS: M. J. Mueller, W. Brodschelm, Anal. Biochem. 218, 425 (1994). Resoln of racemate: R. Kramell et al., Phytochem. Anal. 7, 209 (1996). Review of role in plant development: Y. Koda, Int. Rev. Cytol. 135, 155-199 (1992); of biochemisty and physiology: G. Sembdner, B. Parthier, Annu. Rev. Plant Physiol. Plant Mol. Biol. 44, 569-589 (1993); of signaling in plants: P. Staswick, Curr. Top. Plant Physiol. 11, 14-23 (1993).