5342. Lactobionic Acid
Nomenclature
CAS number: 96-82-2
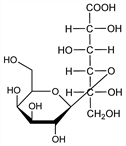
4-O-β-d-Galactopyranosyl-d-gluconic
acid; 4-(β-d-galactosido)-d-gluconic acid. C12H22O12; mol wt 358.30.
C 40.23%, H 6.19%, O 53.58%.
Description and references
Obtained by oxidation of lactose: Fischer, Meyer, Ber. 22, 362 (1889); Ruff, Ollendorff, ibid. 33, 1806 (1900); Isbell, J. Res. Natl. Bur. Stand. 11, 713 (1933); Margariello, US 2746916 (1956 to Nat. Dairy Res. Labs.); Eddy, Nature 181, 904 (1958); Nishizuka et al., J. Biol. Chem. 235, PC13 (1960). Manuf from lactose: Y. Sato et al., DE 2038230 (1971 to Hayashibara Co.), C.A. 74, 142296c (1971). Crystal structure of calcium salt: W. J. Cook, C. E. Bugg, Acta Crystallogr. B29, 215 (1973). NMR studies: T. Taga et al., Bull. Chem. Soc. Jpn. 51, 2278 (1978). For therapeutic use see Erythromycin Lactobionate.
Properties
Syrup. Freely sol in water, slightly sol in methanol, ethanol, glacial acetic acid. Dehydration by distillation with dioxane yields lactobionic δ-lactone, C12H20O11, non-deliquescent crystals, dec 195-196°. Shows mutarotation. [α]D20 +53.0° initial (c = 8.8) → [α]D20 +22.6° final (240 minutes).Derivative
Calcium salt.Nomenclature
Calcium lactobionate.C24H42CaO24; mol wt 754.65.
C 38.20%, H 5.61%, Ca 5.31%, O 50.88%.