5453. Leucomycins
Nomenclature
Description and references
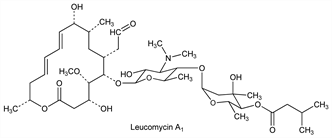
Macrolide antibiotic complex similar to carbomycin, q.v. and erythromycin, q.v., produced by Streptomyces kitasatoensis Hata: T. Hata et al., J. Antibiot. 6A, 87 (1953); Sano, ibid. 7A, 93 (1954). Early work performed on a mixture of at least six components, A1, A2, B1-B4, of which A1 was the most biologically active: J. Abe et al., J. Chem. Soc. Jpn. Pure Chem. Sect. 81, 969 (1960); T. Watanabe et al., Bull. Chem. Soc. Jpn. 33, 1100, 1104 (1960). Leucomycin isolated from an improved strain of S. kitasatoensis is a mixture of at least eight biologically active components, A1 and new substances A3-A9: Hata et al., US 3535309 (1970 to Kitasato Institute and Toyo Jozo). Leucomycins A1, A3-A9, U and V are substituted variations of the structure represented below. Structure of A1: T. Watanabe et al., Angew. Chem. 76, 792 (1964); T. Hata et al., Chem. Pharm. Bull. 15, 358 (1967); of A3: S. Omura et al., Tetrahedron Lett. 1967, 609, 1267; of A4 through A9: eidem, J. Antibiot. 20A, 234 (1967); 21, 272 (1968). Conformation: eidem, Tetrahedron 28, 2839 (1972). Revised configuration at C-9: L. A. Freiberg et al., J. Org. Chem. 39, 2474 (1974). Liquid chromatography of A1, A3-9, U, V: S. Omura et al., J. Antibiot. 26, 795 (1973). Antibacterial and antimycoplasmal activity: Iwata, Akiba, ibid. 15A, 258 (1962); Omura et al., ibid. 21, 532 (1968); 25, 105 (1972). In vitro and preliminary clinical studies: B. C. Stratford, S. Dixson, Med. J. Aust. 1, 1029 (1974). Antimicrobial transformation of A5 to V: K. Singh, S. Rakhit, ibid. 32, 78 (1979). Biosynthesis: C. Kitao et al., ibid. 32, 1055 (1979); S. Omura et al., ibid. 36, 611 (1983). Reviews: Desvignes, Ann. Pharm. Fr. 21, 569 (1963); Toju, Omura, “Chemical and Biological Studies on Leucomycins (Kitasamycins)” in Drug Action and Drug Resistance in Bacteria vol. I, S. Mitsuhashi, Ed. (University Park Press, Baltimore, 1971) pp 267-291; Keller-Schierlein in Fortschr. Chem. Org. Naturst. 30, 313-460 (1973); S. Omura, A. Nakagawa, J. Antibiot. 28, 401-433 (1975).
Derivative
Leucomycin A1, A2, B1-B4 complex.Properties
(Sano, loc. cit.). Powder from acetone; stable at room temp; dec 125°. [α]D20 67.1° (ethanol). uv max (ethanol): 232, 285 nm (E1%1cm 228, 8.6). Freely sol in most organic solvents. Insol in petr ether. Slightly sol in water.Derivative
Leucomycin A3-A9 complex.Properties
White powder; mp 128-145°. uv max (methanol): 231 nm (E1%1cm 353). [α]D25 53° (c = 1 in chloroform). Sol in methanol, ethanol, ethyl acetate, butyl acetate, acetone, benzene, and chloroform; difficultly sol in water. Insol in petr ether. LD50 in mice (mg/kg): >650 i.v., >1000 orally (US 3535309).Derivative
Leucomycin A1.Properties
[α]D25 66.0° (CHCl3). uv max (methanol): 232 nm (E1%1cm 400). pKa′ (50% ethanol): 6.69.Derivative
Triacetylleucomycin A1.Properties
Crystals, mp 125-126°. [α]D25 82.5° (c = 1.3 in CHCl3).Derivative
Leucomycin A3 see Josamycin.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Macrolides