3681. Erythromycin
Nomenclature
Description and references
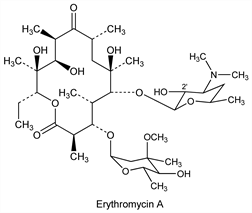
Antibiotic substance produced by a strain of Streptomyces erythreus (Waksman) Waksman & Henrici, found in a soil sample from the Philippine Archipelago. Isoln: McGuire et al., Antibiot. Chemother. 2, 281 (1952); Bunch, McGuire, US 2653899 (1953 to Lilly); Clark, Jr., US 2823203 (1958 to Abbott). Properties: Flynn et al., J. Am. Chem. Soc. 76, 3121 (1954). Solubility data: Weiss et al., Antibiot. Chemother. 7, 374 (1957). Structure: Wiley et al., J. Am. Chem. Soc. 79, 6062 (1957). Configuration: Hofheinz, Grisebach, Ber. 96, 2867 (1963); Harris et al., Tetrahedron Lett. 1965, 679. There are three erythromycins produced during fermentation, designated A, B, and C; A is the major and most important component. Erythromycins A and B contain the same sugar moieties, desosamine, q.v., and cladinose (3-O-methylmycarose). They differ in position 12 of the aglycone, erythronolide, A having an hydroxyl substituent. Component C contains desosamine and the same aglycone present in A but differs by the presence of mycarose, q.v., instead of cladinose. Structure of B: P. F. Wiley et al., J. Am. Chem. Soc. 79, 6070 (1957); of C: eidem, ibid. 6074. Synthesis of the aglycone, erythronolide B: E. J. Corey et al., ibid. 100, 4618, 4620 (1978); of erythronolide A: eidem, ibid. 101, 7131 (1979). Asymmetric total synthesis of erythromycin A: R. B. Woodward et al., ibid. 103, 3215 (1981). NMR spectrum of A: D. J. Ager, C. K. Sood, Magn. Reson. Chem. 25, 948 (1987). HPLC determn in plasma: W. Xiao et al., J. Chromatogr. B 817, 153 (2005). Biosynthesis: Martin, Goldstein, Prog. Antimicrob. Anticancer Chemother., Proc. 6th Int. Congr. Chemother. II, 1112 (1970); Martin et al., Tetrahedron 31, 1985 (1975). Cloning and expression of clustered biosynthetic genes: R. Stanzak et al., Biotechnology 4, 229 (1986). Reviews: T. J. Perun in Drug Action and Drug Resistance in Bacteria 1, S. Mitsuhashi, Ed. (University Park Press, Baltimore, 1977) pp 123-152; Oleinick in Antibiotics vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 396-419; Infection 10, Suppl. 2, S61-S118 (1982). Comprehensive description: W. L. Koch, Anal. Profiles Drug Subs. 8, 159-177 (1979).
Properties
Hydrated crystals from water, mp 135-140°, resolidifies with second mp 190-193°. Melting point taken after drying at 56° and 8 mm. [α]D25 -78° (c = 1.99 in ethanol). uv max (pH 6.3): 280 nm (ε 50). pKa1 8.8. Basic reaction. Readily forms salts with acids. Soly in water: ≈2 mg/ml. Freely sol in alcohols, acetone, chloroform, acetonitrile, ethyl acetate. Moderately sol in ether, ethylene dichloride, amyl acetate.Derivative
Ethylsuccinate.Nomenclature
Description and references
Prepn: GB 830846; R. K. Clark, US 2967129 (1960, 1961 both to Abbott).
Properties
Hydrated crystals from acetone + water, mp 109-110°. [α]D -42.5°.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Macrolides