5466. Levomethadyl Acetate
Nomenclature
Description and references
Longest-acting enantiomer of methadyl acetate, q.v. Duration of action due to active metabolites. Prepn: A. Pohland et al., J. Am. Chem. Soc. 71, 460 (1949). Synthesis of metabolites: F. I. Carroll et al., J. Org. Chem. 41, 3521 (1976). Metabolism in rats: G. L. Henderson et al., Drug Metab. Dispos. 5, 321 (1977); M. Man et al., ibid. 8, 55 (1980); in man: B. S. Finkle et al., J. Anal. Toxicol. 6, 100 (1982). Determn of LAAM and metabolites by HPLC: C.-H. Kiang et al., J. Chromatogr. 222, 81 (1981); by GLC: K. Verebey et al., ibid. 343, 339 (1985). Analgesic activity in mice: N. B. Eddy et al., J. Org. Chem. 17, 321 (1952); in man: A. S. Keats, H. K. Beecher, J. Pharmacol. Exp. Ther. 105, 210 (1952). Pharmacology in monkeys: S. J. Mule, A. L. Misra, Ann. N.Y. Acad. Sci. 311, 199 (1978). Comparison with methadone in the treatment of heroin addiction: T. J. Crowley et al., Psychopharmacology 86, 458 (1985). Review of use in treatment of narcotic addiction: J. D. Blaine et al., Ann. N.Y. Acad. Sci. 362, 101-115 (1981).
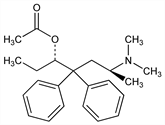
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol-ether, mp 215°. [α]D25 60° (c = 0.2). Sol in water. LD50 in mice (mg/kg): 110.0 s.c., 172.8 orally (Eddy).Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
In treatment of narcotic addiction.