Nomenclature
CAS number: 509-74-0
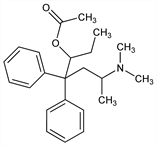
β-[2-(Dimethylamino)propyl]-α-ethyl-β-phenylbenzeneethanol
acetate (ester); 6-(dimethylamino)-4,4-diphenyl-3-heptanol acetate (ester);
O-acetyl-6-dimethylamino-4,4-diphenyl-3-heptanol; 3-acetoxy-6-dimethylamino-4,4-diphenylheptane; 5-acetoxy-2-dimethylamino-4,4-diphenylheptane; acetylmethadol; acemethadone; amidolacetate; race-acetylmethadol.
C
23H
31NO
2; mol wt 353.50.
C 78.15%, H 8.84%, N 3.96%, O 9.05%.
Description and references
Congener of methadone, q.v. Prepn of β-dl-form: M. Bockmühl, G.
Ehrhart, Ann. 561, 52
(1948); of α-dl-form: M. E. Speeter et al., J. Am. Chem. Soc. 71, 57 (1949); R. L. Clark, US 2565592; US 2668814 (1951, 1954 to Merck & Co.); M. E. Speeter, US 2649445 (1953 to Bristol); of α-d and α-l-forms: A. Pohland et al., J. Am. Chem. Soc. 71, 460 (1949). The α-dl-form is more active and less toxic than the β-dl-form. The α-l-form, levomethadyl
acetate, q.v. is less active than the α-d-form but is longer acting. Metabolism of the α-dl-form: R. E. McMahon et al., J.
Pharmacol. Exp. Ther. 149, 436 (1965). Analgesic,
depressant activity of α-dl-form in mice, rats: N. B.
Eddy et al., ibid. 98, 121 (1950); of enantiomers: N. B. Eddy et al., J. Org. Chem. 17, 321 (1952). Comparison of methadone with α-dl-form in
the treatment of heroin addiction: J. H. Jaffe et al., J. Am. Med. Assoc. 211, 1834 (1970).
Derivative
α-dl-Form hydrochloride.
C
23H
32ClNO
2; mol wt 389.96.
C 70.84%, H 8.27%, Cl 9.09%, N 3.59%, O 8.21%.
Properties
Crystals from ethyl acetate, mp 213-214°. Sol in water. pH 4-5.
LD50 in mice (mg/kg): 61.0 s.c., 118.3 orally (Eddy, 1952).Derivative
α-dl-Form hydrobromide.
C
23H
32BrNO
2; mol wt 434.41.
C 63.59%, H 7.42%, Br 18.39%, N 3.22%, O 7.37%.
Properties
Crystals, mp 193-194.5°. Sol in water.Derivative
α-d-Form.
Nomenclature
Alphacetylmethadol. Derivative
α-d-Form hydrochloride.
Properties
Crystals from ethanol-ether, mp 215°. [α]D25 +61.2° (c = 0.2). Sol in water. LD50 in mice (mg/kg): 72.2 s.c., 130.4 orally (Eddy, 1952).Derivative
β-l-Form.
Nomenclature
Betacetylmethadol. Derivative
β-dl-Form hydrochloride.
Properties
Crystals, mp 215-217°. Sol in water. LD50 in mice (mg/kg): 42.0 s.c., 80.2 orally (Eddy, 1952).Note
This is a controlled substance (opiate):
21 CFR, 1308.11.