5468. Levopimaric Acid
Nomenclature
CAS number: 79-54-9
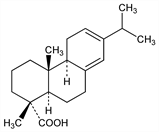
[1R-(1α,4aβ,4bα,10aα)]-1,2,3,4,4a,4b,5,9,10,10a-Decahydro-1,4a-dimethyl-7-(1-methylethyl)-1-phenanthrenecarboxylic
acid; 13-isopropylpodocarpa-8(14),12-dien-15-oic acid; Δ6,8(14)-abietadienoic acid; l-pimaric acid; β-pimaric acid; l-sapietic acid. C20H30O2; mol wt 302.45.
C 79.42%, H 10.00%, O 10.58%.
Description and references
Isolation from American pine oleoresin: Palkin, Harris, J. Am. Chem. Soc. 55, 3677 (1933); from French galipot, from Pinus maritima: Ruzicka, Bacon, Helv. Chim. Acta 20, 1542 (1937); from Pinus palustris: Harris, Sanderson, J. Am. Chem. Soc. 70, 334, 3671 (1948). Structure: Ruzicka, Kaufmann, Helv. Chim. Acta 23, 1346 (1940): cf. Arbuzov, Chem. Zentralbl. 1942, II, 893. Stereochemistry: Schuller, Lawrence, J. Am. Chem. Soc. 83, 2563 (1961); Burgstahler et al., ibid. 83, 4660 (1961); Weiss et al., Chem. Ind. (London) 1962, 1286; Dauben, Coates, J. Org. Chem. 28, 1698 (1963). Conformation: Burgstahler et al., Chem. Commun. 1971, 121; Weiss et al., ibid. 1972, 17.
Properties
Orthorhombic crystals, mp 150°; [α]D20 280.4° (c = 0.7 in alcohol); [α]D14 266.6° (c = 0.4 in chloroform). Absorption max 273 nm: Kraft, Ann. 520, 133 (1935). Practically insol in water, sol in most organic solvents. Forms a crystalline ammonium salt.Derivative
Methyl ester.C21H32O2; mol wt 316.48.
C 79.70%, H 10.19%, O 10.11%.
Properties
Crystals from methanol, mp 64°. [α]D20 268° (c = 1 in ether).Derivative
Molecular compd with quinone.C26H34O4; mol wt 410.55.
C 76.06%, H 8.35%, O 15.59%.